Video Article Open Access
Synthesis and Properties of g-C3N4/Nanocellulose Composite Hydrogels Driven by Light
Xiao-Feng Sun1,2,*, Yangyang Xie1,3, Tao Zhang1, Yudan Li1,3
1Northwestern Polytechnical University, Xian, 710072, China
2Shenzhen Research Institute of Northwestern Polytechnical University, Shenzhen, 518057, China
3Dongguan Sanhang Military Civilian Integration Innovation Research Institute, Dongguan, 523808, China
Vid. Proc. Adv. Mater., Volume 3, Article ID 2210369 (2022)
DOI: 10.5185/vpoam.2022.10369
Publication Date (Web): 26 Nov 2023
Copyright © IAAM
Graphical Abstract
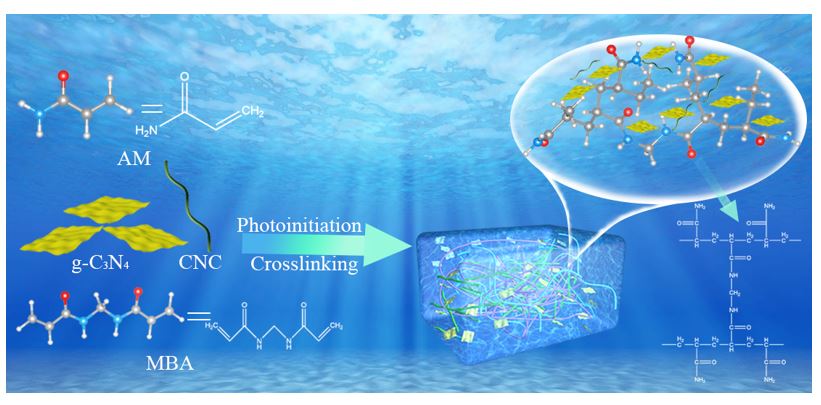
Abstract
Recently, the synthesis of photoactive hydrogels using g-C3N4 as initiator has attracted more and more attentions. In this paper, g-C3N4/CNC/PAM composite hydrogel was prepared under visible light and the morphology and structure of the prepared hydrogel were characterized. The swelling properties and the swelling mechanism of g-C3N4/CNC/PAM composite hydrogel were studied, and the hydrogels had pH sensitivity, and the equilibrium swelling rate of the hydrogels increased with the increased of the pH value. The equilibrium swelling of the hydrogels conformed to the Fickian swelling behaviour at the initial stage, and the swelling of the hydrogels conformed to the Schott second-order kinetic model in the whole swelling process. With an increase in the content of each component in the hydrogel, the hardness of the hydrogel was enhanced but the toughness was weakened. The maximum compression strength and fracture compression deformation rate of the hydrogel were 542 kPa and 89%, respectively. The photocatalytic degradation properties and the photocatalytic degradation kinetics of methylene blue (MB) and rhodamine B by g-C3N4/CNC/PAM composite hydrogels were studied. The results showed that the hydrogels had good photocatalytic degradation ability for both MB and RhB, and the pH of solution, g-C3N4 content, MB initial concentration and initial quality of the hydrogels can all affect the photocatalytic degradation ability of the hydrogels. When the initial concentration of MB and the mass of hydrogel were 400 mg/L and 0.02 g, respectively, the photocatalytic degradation of methylene blue reached the maximum value of 881 mg/g at the pH=11.00. When the initial concentration of RhB and the initial mass of the hydrogel were 100 mg/L and 0.02 g, respectively, the photocatalytic degradation of RhB by the hydrogel reached the maximum value of 110 mg/g at the pH=11.00. In addition, the photocatalytic degradation of MB by the hydrogels conforms to the Langmuir-Hinshelwood first-order kinetic model, while the photocatalytic degradation of RhB by the hydrogels is more consistent with the Langmuir-Hinshelwood zero-order kinetic model. Finally, the reusability and photocatalytic degradation mechanism of g-C3N4/CNC/PAM composite hydrogels were studied. The results of repeated experiments showed that the prepared hydrogel was reusable, and its photocatalytic degradation rate of MB and RhB didn’t decrease significantly even after the reuse of five times. The results of free radical capture experiment showed that the photocatalytic degradation of MB and RhB by the hydrogel was mainly realized by two kinds of free radicals (•OH and •O2-).
Keywords
Graphite phase carbon nitride; Cellulose nanocrystals; Hydrogels; Photocatalytic degradation.
Acknowledgement
This research was funded by the Shenzhen Science and Technology Project for Sustainable Development (No. KCXFZ20201221173004012) and the National College Students Innovation and Entrepreneurship Training Program (202110699137).
References
- Y. Guo, J. Bae, Z. Fang, P. Li, F. Zhao, G. Yu, Chemical Reviews, 2020, 120(15), 7642.
- X.-F. Sun, Z. Gan, Z.X. Jing, H.H. Wang, D. Wang, Y.N. Jin, Journal of Applied Polymers Science, 2015, 132(10), 41606.
Biography
Xiao-Feng Sun obtained his PhD degree at University of Bangor of United Kingdom in 2006, and then, as a research officer, he worked on an industrially based project involving the use of a mixed culture of microbes to produce a specific synthon for the pharmaceutical industry. In 2007, he came back to China, and focused his study on environmental materials at Zhejiang University. Now he worked at Northwestern Polytechnical University, and he has published more than 100 papers which have been cited more than 5000 times by other scientists. His research concerns biomass-based materials and nanomaterials for environmental protection.
Video Proceedings of Advanced Materials

Upcoming Congress



