Video Article Open Access
The Chemistry of Reticular Materials for Carbon Capture and Conversion
Kyle E. Cordova1,2,3
1Materials Discovery Research Unit, Advanced Research Centre, Royal Scientific Society, Amman 11941, Jordan
2Department of Chemistry, Faculty of Science, Universiti Putra Malaysia (UPM), Selangor 43400, Malaysia
3Foundry of Reticular Materials for Sustainability (FORMS), Materials Synthesis and Characterization Laboratory, Institute of Advanced Technology, UPM, Selangor 43400, Malaysia
Vid. Proc. Adv. Mater., Volume 3, Article ID 2203254 (2022)
DOI: 10.5185/vpoam.2022.03254
Publication Date (Web): 22 Jul 2022
Copyright © IAAM
Graphical Abstract
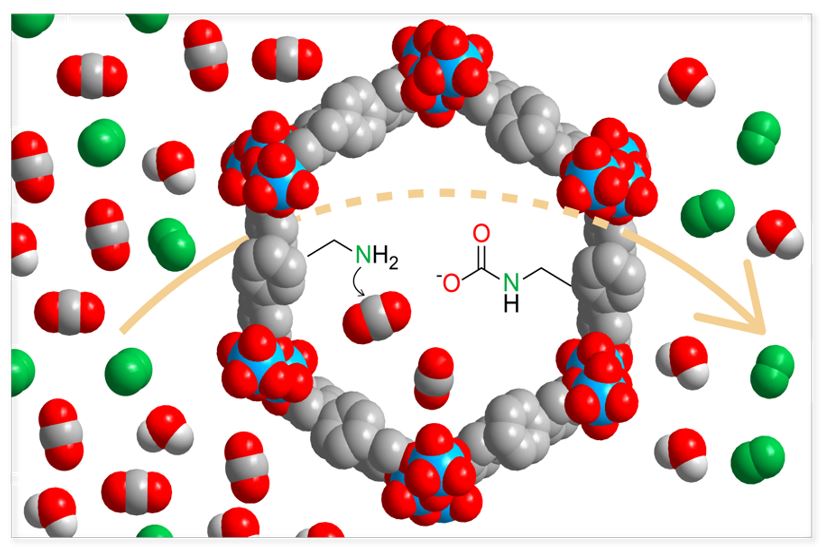
Abstract
The carbon dioxide challenge is one of the most pressing problems facing our planet. Each stage in the carbon cycle — capture, regeneration and conversion — has its own materials requirements. Metal-organic frameworks (MOFs) are a class of crystalline, porous reticular materials, where organic linkers and inorganic units are alternately connected to form open, extended frameworks. The flexibility with which these components can be varied has led to the most extensive class of materials with high surface areas and high thermal/chemical stability. These characteristics allow the interior of MOFs to be chemically altered for use in gas storage, gas separation, and catalysis, among many other applications. The precise control over the assembly of MOFs is expected to propel this field further into new realms of synthetic chemistry in which far more sophisticated materials may be accessed in order to more effectively address the global challenge of climate change resulting partially from uncontrolled anthropogenic CO2 emission. In this seminar, I will discuss our success in rationally and systematically modulating the interplay between the structures of reticular materials, such as MOFs, and the desired output chemical properties in order to achieve exceptionally selective capture and effective catalytic conversion of CO2 to value-added products. I will demonstrate how the interior of reticular materials can be designed with a level of precision that is crucial for the development of the next generation of CO2 adsorbents as well as higher-performing catalysts for CO2 conversion. To realize a total solution, I will argue the case that the precision of reticular chemistry is essential for building more complex materials to address selectivity, capacity and conversion together in one material.
Keywords
Reticular chemistry; metal-organic frameworks; porous materials; carbon dioxide capture and conversion.
Acknowledgement
K.E.C. is grateful for financial support from the Jordan Ministry of Higher Education and Scientific Research (No. BAS/1/6/2020) and the Newton-Khalidi Fund and the Royal Academy of Engineering’s Transforming Systems through Partnership program (No. TSP2021/100001).
References
- X. Yao, K. E. Cordova, Y.-B. Zhang, Flexible Metal-Organic Frameworks En Route to Energy-Efficient Carbon Capture, Small Structures, 2022, DOI: 10.1002/sstr.202100209.
- M. M. Abdelnaby, K. E. Cordova, I. Abdulazeez, A. M. Alloush, B. A. Al-Maythalony, Y. Mankour, K. Alhooshani, T. A. Saleh, O. C. S. Al Hamouz, Novel Porous Organic Polymer for the Concurrent and Selective Removal of Hydrogen Sulfide and Carbon Dioxide from Natural Gas Streams, ACS Applied Materials and Interfaces, 2020, 12, 47984.
- C. S. Diercks, Y. Liu, K. E. Cordova, O. M. Yaghi, The Role of Reticular Chemistry in the Design of CO2 Reduction Catalysts, Nature Materials, 2018, 17, 301.
- C. A. Trickett, A. Helal, B. A. Al-Maythalony, Z. H. Yamani, K. E. Cordova, O. M. Yaghi, The Chemistry of Metal-Organic Frameworks for CO2 Capture, Regeneration, and Conversion. Nature Reviews Materials, 2017, 2, 17045.
- H. Furukawa, K. E. Cordova, M. O’Keeffe, O. M. Yaghi, The Chemistry and Applications of Metal-Organic Frameworks. Science, 2013, 341, 1230444.
Biography
Kyle E. Cordova is Executive Director for Scientific Research and Senior Assistant to Her Royal Highness Princess Sumaya bint El Hassan for Scientific Affairs at the Royal Scientific Society, Jordan and an Adjunct Professor in the Department of Chemistry at the Universiti Putra Malaysia, Malaysia. Kyle received his graduate degree in inorganic chemistry from the University of California, Los Angeles (UCLA) and undergraduate degree in chemistry from the University of San Diego. He was a founding leader of the University of California, Berkeley Global Science Institute (BGSI) – an organization whose mission is to partner with institutions of learning, foundations, governments, and industry to create centers of research in the foundational sciences that address global problems and build a culture of global science. During his tenure at BGSI, Mr. Cordova was responsible for designing, implementing, and managing all global science centers in the United States and abroad (Japan, South Korea, Saudi Arabia, United Arab Emirates, Kuwait, Jordan, China, Malaysia, Indonesia, Argentina, and Mexico).
Kyle’s research continues to focus on further developing and promoting the principles of reticular chemistry for applications in clean energy, health, and the environment. He has co-authored more than 40 publications, including 5 in Science or Nature family journals, with >16,000 citations. Kyle has received numerous awards including being named a Leader in Innovation Fellow of the Newton Fund and UK Royal Academy of Engineering, International Union of Pure and Applied Chemistry (IUPAC) U.S. Young Observer for the US National Academies of Sciences, Engineering, and Medicine, Ronald E. McNair Scholar, and US National Cancer Institute CURE Scholar. He has recently founded a spin-off company, AquaPoro Ventures, to commercialize his research discoveries for water applications. AquaPoro Ventures won the Queen Rania National Entrepreneurship Competition and UNICEF Water, Sanitation, and Hygiene Entrepreneurship Competition in 2021.
Video Proceedings of Advanced Materials

Upcoming Congress



