Video Article Open Access
Calcium-Rich Compound in Concrete as Filler, Activator, and CO2 Capturing-Storing Function towards Combating Climate Change
Nur Hafizah A. Khalid*, Adrina Rosseira Abu Talip, Nur Nadhira Abdul Rasid
Universiti Teknologi Malaysia,81310 Johor Bahru, Malaysia
Vid. Proc. Adv. Mater., Volume 3, Article ID 2203252 (2022)
DOI: 10.5185/vpoam.2022.03252
Publication Date (Web): 23 Jul 2022
Copyright © IAAM
Graphical Abstract
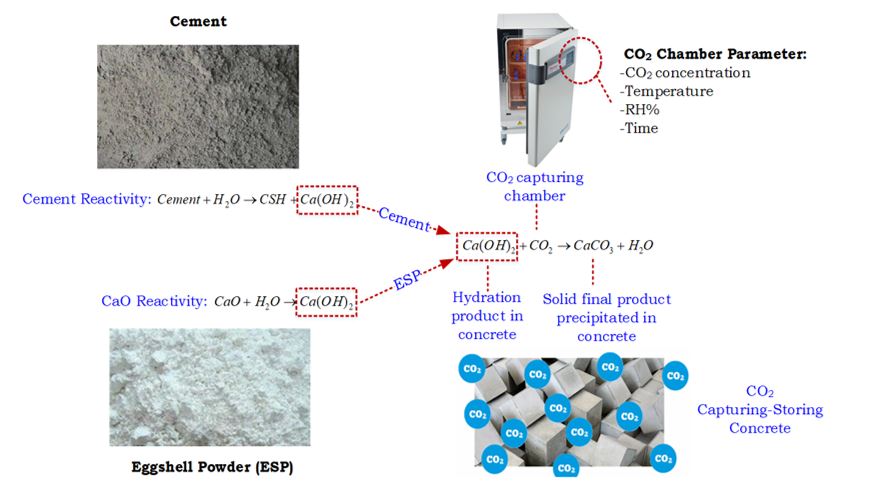
Abstract
A Calcium-rich compound plays a vital role in concrete-making depending on the intended usage. An example is the calcium carbonate (CaCO3) - a common concrete filler. Quick lime or calcium oxide (CaO), on the other hand, has different ‘acting energy’ in cementitious system. The selection depends predominantly on the application. If the intention is to produce precast concrete product or for rapid construction project, then early concrete strength development is pivotal. However, the process usually compromises the shrinkage and porosity of the concrete. This has made blended cement concrete more preferable, even though its curing time is twice as long as ordinary Portland cement due to its low thermal conductivity. The long curing period was eventually resolved by adding accelerator into the concrete mix. The research has shown that these accelerators can be produced from green or sustainable material that are easily available. Such accelerator not only speeds up the curing time, it also contributes towards the preservation of natural resources. A good green accelerator is the bio-energized lime. This is produced from sustainable and modified sources to transform CaCO3 into CaO through calcination at a pre-set temperature. An example raw material is the eggshell in powder form. This is commonly available as an aviculture waste and contains more than 90% CaCO3 that are convertible into cementitious material. When the CaCO3 is calcined at a certain high temperature, it is transformed into CaO and the changes are determined under microstructure examination. Here, the function of the calcium in a cementitious system is changed from filler to activator, depending on the CaCO3-CaO transformation energy. For better identification on the cycle function of the calcium rich compound, an experiment was conducted by curing a series of concrete samples with CaO content in an enclosed chamber infiltrated with CO2 gas, termed as the “CO2 curing method”. Results shown that the Ca(OH)2-CO2 reactivity in the concrete was transformed into a solid calcium carbonate (CaCO3) to fill the concrete pore, making the concrete denser and more durable. It also became clear that the process had induced the carbon capturing and storing (CCS) function through a chemical reaction to store CO2 permanently in the form of thermodynamically stable carbonates. To conclude, the current research intends to enrich the current knowledge bank and contribute towards sustainable development in the following manner - (i) to reduce unsustainable waste disposal at landfill sites; (ii) to reduce the amount of clinker required to produce unsustainable cement; (iii) to reduce CO2 emission from the construction industry; (iv) to combat climate change in the long run via a more sustainable and intelligent production of CO2 capturing concrete; and finally (v) to advocate the conversion of any allied waste material with similar characteristics for the development of smart and sustainable concrete material.
Keywords
Calcium carbonate; calcium oxide; calcium hydroxide; carbon dioxide; bio-lime.
Acknowledgement
The financial support for this project is provided by the Ministry of High Education Malaysia (MoHE) and Universiti Teknologi Malaysia (UTM) under the grant R.J130000.7851.5F034, Q.J130000.3651.03M24, and Q.J130000.2451.09G91. The authors are also appreciative to the Talented Young Scientist Program (TYSP) for sponsoring the mobility fund for Dr. Nur Hafizah A. Khalid to Hunan University, China which provides her access to their facilities as well as relevant support for the experimental works.
References
- Mo, K. H.; Alengaram, U.J.; Jumaat, M.Z.; Lee, S.C.; Goh, W.I.; Yuen, C.W.; Recycling of seashell waste in concrete: A review. Constr. Build. Mater., 2018, 162, 751-764.
- Pliya, P.; Cree. D.; Limestone derived eggshell powder as a replacement in Portland cement mortar. Constr. Build. Mater., 2015, 95, 1-9.
- Galván-Ruiz, M.; Hernández, J.; Baños, L.; Noriega-Montes, J.; Rodríguez-García, M.E.; Characterization of calcium carbonate, calcium oxide, and calcium hydroxide as starting point to the improvement of lime for their use in construction, J. Mater. Civ. Eng., 2009, 21(11), 694-698.
- Qian, X.; Wang, J.; Fng, Y.; Wang, L.; Carbon dioxide as an admixture for better performance of OPC-based concrete,” J. CO2 Util., 2018, 25, 31-38.
- Abdul, M.; Naganathan, S.; Abdul, H.; Nasharuddin, K.; Performance of lime kiln dust as cementitious material, Procedia Eng., 2015, 125, 780-787.
Biography
Nur Hafizah Abd Khalid is a senior lecturer at School of Civil Engineering, Universiti Teknologi Malaysia (UTM) and a research member of Construction Material Research Group (CMRG). Previously, she has been appointed as Council Member of Concrete Society Malaysia (CSM) for 2 years. She completed all degrees in Universiti Teknologi Malaysia (UTM) and completed her PhD in year 2016. Along her career and research journey, she experienced receiving prestigious international awards such as Young Women Scientist 2014 (Korea), Talented Young Scientist 2018-2019 (China), the ASEAN-ROK Next Innovator 2020 (Korea) and prestigious local award of UTMShine 2021 (Malaysia). Her research interest focuses on Advanced Concrete Materials, Green and Environmental-Friendly Concrete, Nano Materials, Characterization of Materials, Chemical Dynamics of Materials. Her biggest interest is in recycling wastes into value-added concrete material which to promote low-embodied-energy construction material and to bridge the gap between research innovation and industrial application.
Video Proceedings of Advanced Materials

Upcoming Congress



