Video Article Open Access
Microfluidic Elaboration of Polymeric Micro-carriers with Complex Morphologies for New Drug Delivery Strategies
Ikram Ullah Khan1,2, Christophe A. Serra1*, Nicolas Anton2, Marc Schmutz1, Isabelle Kraus3, Thierry F. Vandamme2
1Université de Strasbourg, CNRS, ICS UPR 22, 25 rue Becquerel, 67034 Strasbourg, France
2Université de Strasbourg, CNRS, CAMB UMR 7199, 74 Route du Rhin, 67401 Illkirch, France
3Université de Strasbourg, CNRS, IPCMS UMR 7504, 23 rue du Loess, 67034 Strasbourg, France
Vid. Proc. Adv. Mater., Volume 2, Article ID 2021-02101 (2021)
DOI: 10.5185/vpoam.2021.02101
Publication Date (Web): 20 Mar 2021
Copyright © IAAM
Graphical Abstract
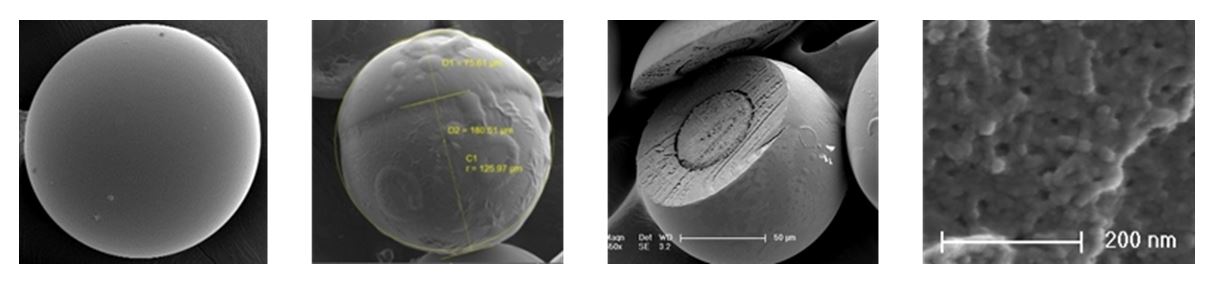
Abstract
Microencapsulation is used for the protection of drug, controlled release, reduced administration frequency, patient comfort and compliance. In comparison with conventional techniques for encapsulation, microfluidics offers a new route to precisely control over microcarriers’ size, shape, morphology, composition and thus release properties.
Continuous-flow off-the-shelves capillaries-based microfluidic droplet generators, assembled within minutes [1], were used to produce size-controlled and drug-loaded plain, core-shell, Janus and Trojan polymeric microcarriers. A single capillary-based device was employed to obtained either poly(ethyl acrylate) plain microparticles [2] or poly(acrylamide) Trojan microparticles embedding with drug-loaded poly(ethyl acrylate) nanoparticles previously obtained from the nanoemulsification of the monomer phase with an elongation-flow micromixer [3]. On the opposite, a two capillaries-based device was employed to prepare poly(acrylamide)/poly(methyl acrylate) core-shell [4] and Janus [5] microparticles encapsulating two incompatible drug in two spate domain.
This lecture proposes to study the production and the release properties of these microcarriers as well as the subsequent new release strategies (e.g., sequential, synergetic, nanoparticle delivery to GIT etc.) arising from these uncommon morphologies. It will be demonstrated how operating parameters (fluids’ flow rate, nature of the monomer, concentration of the surfactant etc.) can affect the size and the morphology of the polymeric microcarriers and how to tune the sustained release of a single drug or two incompatible drugs encapsulated into a single microparticle.
Keywords
Microfluidics, polymer, microparticles, drug, delivery
Acknowledgement
Please acknowledge the funding authorities and other research supports.
References
- Chang Z., C. Serra, M. Bouquey, L. Prat and G. Hadziioannou, Lab. Chip., 2009, 9, 3007-3011.
- Khan I.U., C.A. Serra, N. Anton and T. Vandamme, Int. J. Pharm., 2013, 441 (1), 809-817
- Khan U.K., C.A. Serra, N. Anton, M. Er-Rafik, C. Blanck, M. Schmutz, I. Kraus, N. Messaddeq, C. Sutter, H. Anton, A.S. Klymchenko, T.F. Vandamme, Int. J. Pharm., 2015, 493 (1-2),7-15.
- Khan I.U., L. Stolch, C.A. Serra, N. Anton, R. Akasov and T. Vandamme, Int. J. Pharm., 2015, 478 (1), 78-87
- Khan, I.U., C.A. Serra, N. Anton, X. Li, A. Roman, N. Messaddeq, I. Kraus and T. Vandamme, Int. J. Pharm., 2014, 473 (1–2), 239-249.
Biography
Christophe A. Serra graduated in 1992 from the Engineering School of Chemical Engineering (ENSIC) in Nancy (France). He received his Ph.D. in 1996 from the University Paul Sabatier in Toulouse (France). Then, he spent 18 months as a postdoctoral researcher at Rice University (Houston, TX). Since 1998, he has been a faculty member of the University of Strasbourg (France) teaching chemical engineering at the European Engineering School of Chemistry, Polymers and Materials Science (ECPM). His researches concern the development of new microfluidic-assisted polymer processes for the synthesis of architecture-controlled polymers and functional polymeric micro- and nanoparticles. In 2010 he was appointed Full Professor at the University of Strasbourg and joined in 2015 the Charles Sadron Institute (ICS) becoming one of its deputy directors.
Video Proceedings of Advanced Materials

Upcoming Congress



